Extended Follow-up of HOVON-65/GMMG-HD4 Trial Confirms Superior PFS and OS in Newly Diagnosed MM Patients Administered Bortezomib During Induction and Maintenance Therapy
• Randomized, open-label, phase III trial[1]
Summary of Key Conclusions
• Long-term follow-up of previously published HOVON-65/GMMG-HD4 trial confirms that use of bortezomib-based induction regimen and maintenance therapy significantly improved PFS and OS in newly diagnosed transplant-eligible multiple myeloma (MM) patients compared with patients on vincristine-based induction regimen with thalidomide maintenance
• Bortezomib improved long-term outcomes in patients with renal failure
• Bortezomib improved outcome in patients with intermediate-/poor-risk cytogenetics
• No increased risk of developing second primary malignancies observed with bortezomib
• Fewer patients receiving bortezomib maintenance therapy discontinued due to adverse events vs thalidomide maintenance therapy
Background
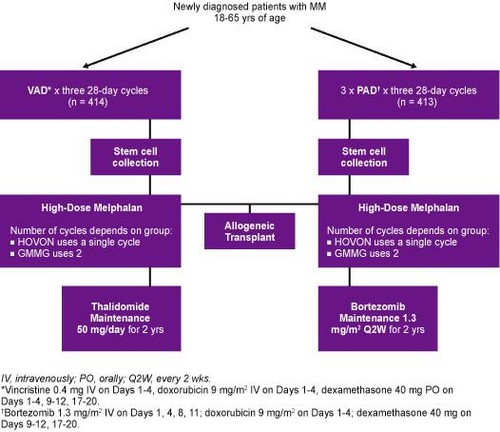
• Previous phase III HOVON-65/GMMG-HD4 trial[2] conducted to evaluate the efficacy of bortezomib as part of induction regimen with doxorubicin and dexamethasone (PAD) and as maintenance therapy vs vincristine/doxorubicin/dexamethasone (VAD) induction regimen with thalidomide maintenance
o Both groups received high-dose melphalan and autologous stem cell transplant following induction
o Integrating bortezomib into induction and maintenance regimen significantly improved PFS vs VAD group
o Subanalysis demonstrated that high-risk subgroups, ie, renal failure and del(17p), achieved superior OS and PFS in patients in PAD arm over patients in VAD arm[2,3]
• Superior outcome of bortezomib confirmed in meta-analysis of 4 phase III trials comparing bortezomib to non-bortezomib regimens[4]
• Current study designed to evaluate whether PFS and OS benefit maintained in extended follow-up of patients in HOVON-65/GMMG-HD4 study
Eligibility
• Main inclusion criteria
o New diagnosis of symptomatic, Salmon-Durie stage II/III, A+B MM
o Stages I, II, or III according to International Staging System
o 18-65 years of age
o World Health Organization performance status 0-3
o Renal failure included
• Exclusion criteria
o Amyloid light chain amyloidosis
o Nonmeasurable or nonsecretory disease
o Baseline neuropathy grades 2-4 in severity
o Severe concomitant disease
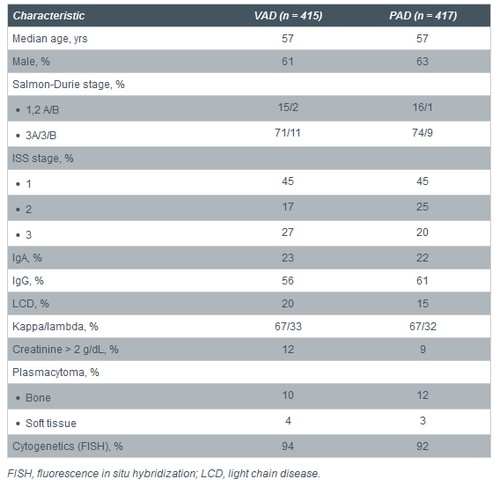
Baseline Characteristics
Description of Current Analysis
• This follow-up study data based on
o 835 patients accrued from May 2005 to June 2008, with 827 deemed eligible
o Final analysis cutoff date: November 2013
• Primary endpoint
o Long-term PFS, allogeneic stem cell transplant censored
• Secondary endpoints
o Response after induction, high-dose maintenance, and on protocol
o Long-term OS from randomization
o Toxicity
o PFS from high-dose melphalan
• Response and relapse/progression evaluation
o European Bone Marrow Transplant criteria
o Near CR:CR by immunofixation (not available or positive)
o Very good PR per International Myeloma Working Group criteria
Main Findings
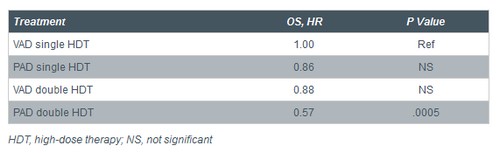
• Bortezomib therapy continued to provide significant PFS (HR: 0.76; 95% CI: 0.64-0.90; P = .001) and OS benefit (HR: 0.78; 95% CI: 0.64-0.90; P = .01) vs VAD
o Median PFS, PAD arm vs VAD arm: 36 vs 27 months, respectively
o Median OS, PAD arm vs VAD arm: not reached vs 84 months, respectively
• Patients with renal failure on PAD arm had significantly improved PFS (HR 0.44; CI 0.26-0.75; P = .003) and OS (HR: 0.38; CI 0.21-0.69; P <.001) vs patients with renal failure on VAD arm
• Bortezomib improved outcome in patients with intermediate-/poor-risk based on FISH/ISS
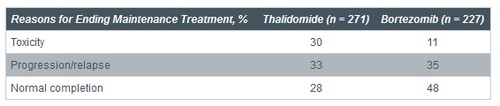
• Lower proportion of patients discontinued maintenance due to toxicities with bortezomib vs thalidomide
Other Outcomes
• Bortezomib treatment shown to be independent prognostic factor in patients with poor-risk FISH (HR: 0.71; 95% CI: 0.52-0.99; P = .035) in landmark analysis for OS from initiation of maintenance therapy
• Bortezomib therapy did not decrease time to developing second primary malignancy (HR: 0.68; 95% CI: 0.38-1.27; P = NS)
• Bortezomib therapy provided most benefit when used with 2 cycles high-dose melphalan therapy and autologous stem cell transplant vs single cycle
References
1. Sonneveld P, Scheid C, van der Holt B, et al. Bortezomib induction and maintenance treatment improves survival in patients with newly diagnosed multiple myeloma: extended follow-up of the HOVON-65/GMMG-HD4 trial. Program and abstracts of the 55th American Society of Hematology Annual Meeting and Exposition; December 7-10, 2013; New Orleans, Louisiana. Abstract 404.
2. Sonneveld P, Schmidt-Wolf IG, van der Holt B, et al. Bortezomib induction and maintenance treatment in patients with newly diagnosed multiple myeloma: results of the randomized phase III HOVON-65/ GMMG-HD4 trial. J Clin Oncol. 2012;30:2946-2955.
3. Scheid C, Sonneveld P, Schmidt-Wolf I, et al. Bortezomib before and after autologous stem cell transplantation overcomes the negative prognostic impact of renal impairment in newly diagnosed multiple myeloma: a subgroup analysis from the HOVON-65/GMMG-HD4 trial. Haematologica. 2013;[Epub ahead of print].
4. Sonneveld P, Goldschmidt H, Rosinol L, et al. Bortezomib-based versus nonbortezomib-based induction treatment before autologous stem-cell transplantation in patients with previously untreated multiple myeloma: a meta-analysis of phase III randomized, controlled trials. J Clin Oncol. 2013;31:3279-3287.





